 What is the ATLANTIS Study?
What is the ATLANTIS Study?
Thank you for your interest in the ATLANTIS Study. The ATLANTIS Study is a multinational clinical research study testing the study medication, amlitelimab, in children and adults 12 years of age and older with moderate to severe atopic dermatitis.
Researchers want to see if the study medication (amlitelimab) can safely treat moderate to severe atopic dermatitis over a long period of time.
Amlitelimab is not yet approved by health authorities such as the US Food and Drug Administration (FDA) and is still under investigation for the treatment of atopic dermatitis, so its effectiveness and safety have not been established.
Click below to download resources containing more information on atopic dermatitis and the study.
Understanding Clinical Trials (for 12 to 17 year olds)
Watch to learn more about the clinical research studies.
Understanding Atopic Dermatitis (for 12 to 17 year olds)
Watch to learn more about atopic dermatitis.

 Who can participate in the ATLANTIS Study?
Who can participate in the ATLANTIS Study?
This study is for children and adults 12 years of age and older:
- Who have had moderate to severe atopic dermatitis for 1 year or longer.
- For whom topical medication is not advised OR
- Whose symptoms did not improve with topical medication in the last 6 months.
Additional participation criteria also apply.
 What study medication will I receive?
What study medication will I receive?
If you qualify for the study, you will receive the study medication, amlitelimab, every 4 weeks for about 3 years. You will receive your final dose at Week 156.
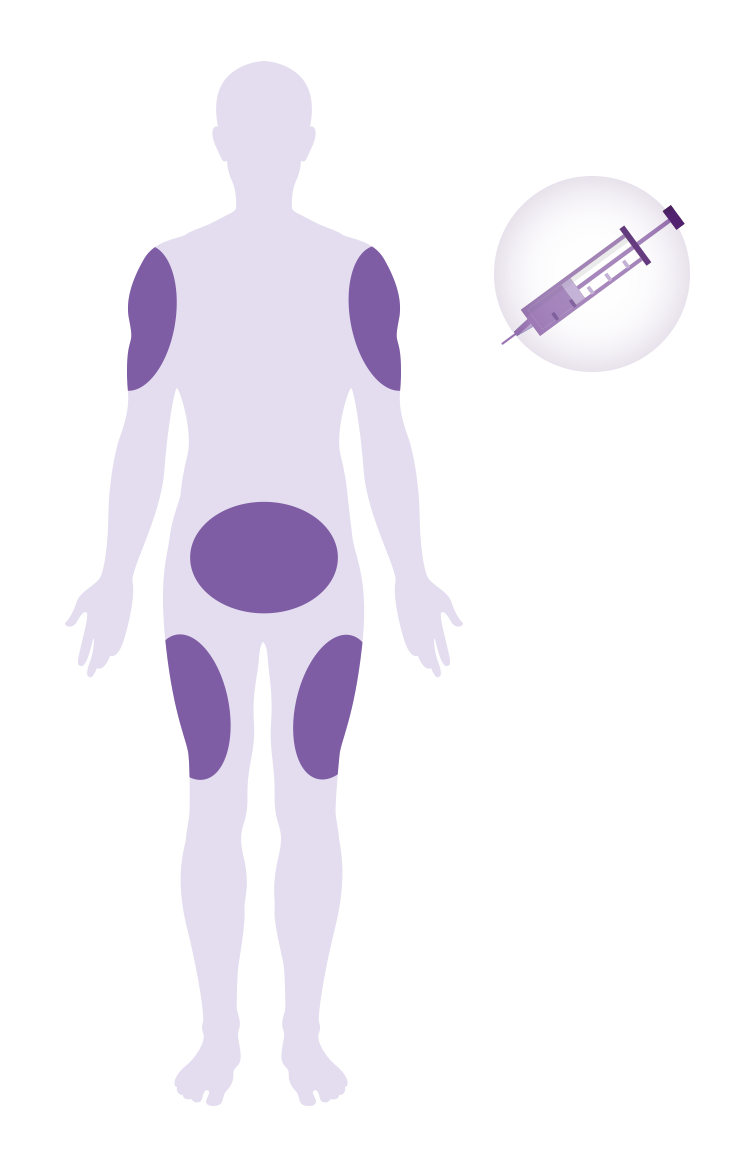
Amlitelimab is given as an injection (shot) into the fatty layer just beneath the skin. It can be given in the abdomen, outer thigh, or upper arm.*
- For the first year (52 weeks) you will receive your dose of amlitelimab at the study site.
- After that, you may choose to continue receiving your injections at the study site or you/your caregiver can be trained to give the injection at home.
During the study you may be able to keep using certain topical medications, moisturizers, and steroid creams. The study doctor will let you know which treatments you are allowed to use.
*Upper arms can only be injected by a caregiver, not by the participant him/herself.
 What happens during the study?
What happens during the study?
First you will need to give your consent, or permission, to join the study before any study procedures can begin by reading and signing an Informed Consent Form. Participants who are 12 to 17 years of age will sign a similar form called the Informed Assent Form in addition to the Informed Consent Form signed by the participant’s legal authorized representative.
The ATLANTIS Study lasts about 3.5 years and is divided into 3 parts. While you are in the study, you will have about 26 visits for tests and health checks.
Screening Period
(lasts 2 to 4 weeks)
To determine whether you qualify for the study, the study team will ask questions about your health and the medicines you take and will run some tests.
Study Treatment Period
Lasts 160 weeks (about 3 years)
You will receive the study medication, amlitelimab, every 4 weeks for about 3 years (160 weeks). You will receive your final dose at Week 156. During the first year you will have visits at Week 0, Week 2, and Week 4, then every 4 weeks until Week 52. For the following 2 years (approximately), you will have visits every 3 months (12 weeks).
Follow-Up Period
20 weeks after the final dose of IMP (about 5 months)
20 weeks after your final dose of amlitelimab, you will have 1 more visit to check on your health.
 What kind of tests and health checks will I have?
What kind of tests and health checks will I have?
During the study you will have about 26 on-site visits for tests and health checks. You will not have all these tests at each visit. There may also be additional tests. Talk to the study doctor for more information.
Physical exam
Height
Weight
Vital signs
Heart activity (ECG)
Blood tests
Urine test
COVID-19 test
Atopic dermatitis severity score
Questionnaires
Side effect check
Mental health check
Is there anything else I will have to do during the study?
You will have an electronic diary (eDiary) app installed on your phone or on a device provided to you. The study team will train you on how to use this app to complete questionnaires. You’ll complete questionnaires every day for the first 6 months, then at different times during the remainder of the study. The study team will let you know when you are expected to complete the questionnaires.